Roundtable Discussion: Factors for Choosing an AR-Targeted Therapy Considered for Prostate Cancer
Alan H. Bryce, MD, the chair of the Genitourinary Disease Group at the Mayo Clinic leads a Targeted Oncology Rountable Discussion on factors for choosing AR-targeted therapy for prostate cancer. He is joined by Robert Yoo, DO, Justin Goldfarb, DO, Miguel Gonzalez Velez, MD, Gehan Botrus, MD, Fei Gu, MD, Rajinder Grover, MD.
Alan H. Bryce, MD


Alan H. Bryce, MD, the chair of the Genitourinary Disease Group at the Mayo Clinic leads a Targeted Oncology Roundtable Discussion on factors for choosing AR-targeted therapy for prostate cancer. He is joined by Robert Yoo, DO, Justin Goldfarb, DO, Miguel Gonzalez Velez, MD, Gehan Botrus, MD, Fei Gu, MD, Rajinder Grover, MD.

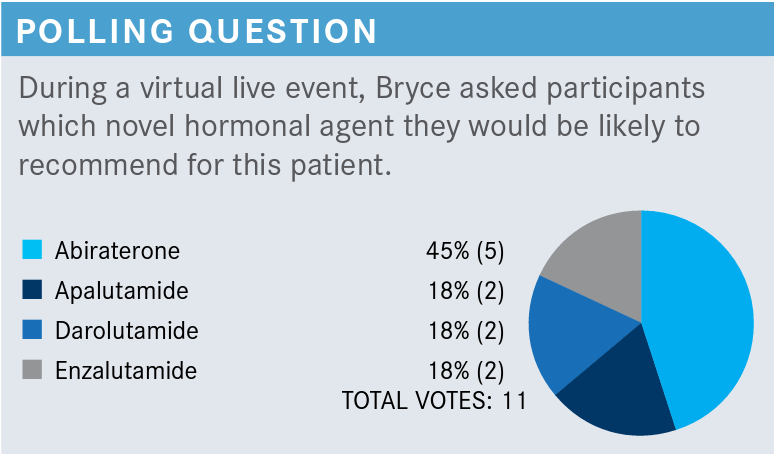
BRYCE: Abiraterone [Zytiga] was clearly the most popular option with 5 votes.
YOO: I voted for apalutamide [Erleada] because of the once-a-day, easy dosing.
BRYCE: Fair enough, practical consideration, I understand that. GOLDFARB: I voted for abiraterone. I have the most experience with it and [have had] no issues with insurance coverage in the past.
BRYCE: Familiarity and availability. All of these agents will have positive efficacy. The PSA will go down. I think the point here, and the insurance company may well push back, but abiraterone is of course approved for metastatic disease, which means visible on imaging. But in the nomenclature of current prostate cancer paradigms, we would refer to this patient as having nonmetastatic castration-resistant disease, which means not localizable by conventional imaging. Now, the issue there of course is maybe in the era of molecular imaging this space will disappear, because suddenly we’ll be able to localize disease much more effectively. But it remains an open question how that patient should be treated since all of the data are based on conventional imaging, not molecular imaging. So technically speaking, at the moment, only options 2, 3, and 4 are approved in the nonmetastatic setting. So that’s where we’re going to focus our discussion.
GONZALEZ: I chose darolutamide [Nubeqa] because there is a comparison for the nonmetastatic setting that showed less toxicity and a better safety profile particularly from the brain-barrier crossing compared to the other ones.1
BOTRUS: Patient already has a seizure.
BRYCE: Yeah, so patient has a seizure history, and that was kind of a key point that was put in there, and we’ll talk about why that differentiates amongst the drugs here.

BRYCE: Dr Botrus, do you want to expand on why the patient’s seizure history makes a difference here?
BOTRUS: So darolutamide doesn’t cross the blood-brain barrier, so it [can] be used for the patients who have a seizure. [It’s a] safe choice to be given. So, darolutamide is one of the choices that also can be given if the patient has seizure activity, as it doesn’t cross the blood-brain barrier.
BRYCE: Of the 3 approved drugs in this setting—apalutamide, enzalutamide [Xtandi], and darolutamide—darolutamide is the 1 that doesn’t cross the blood-brain barrier, has much lower concentrations than the others, and as such, doesn’t have the same CNS [central nervous system] toxicity [as] some of the data [from the other trials of the other agents]. It’s one of the differentiating toxicities amongst these drugs, if you will. There’s a couple others, but this is one of the big ones.
The point about PSA doubling time, does anybody want to answer what [would be different] if the PSA doubling time was 15 months? Is this something you incorporate into your management of these patients or think about?
BOTRUS: It should be 10 months, the doubling time.
BRYCE: All 3 clinical trials required a doubling time of 10 months or faster for enrollment, though, in fact, the data really only apply to those patients, right? I mean, the fact of the matter is, we don’t have data for patients with a PSA doubling time of 15 months, and in that situation, I would say the question is not will the drugs work—mechanistically, they still work—but the question is, do they actually confer a viable advantage? The answer to that is, we don’t know. We simply don’t have the data one way or the other, but technically speaking, everything we’re going to talk about in terms of outcomes data with these drugs is based on a doubling time of less than or equal to 10 months.

BRYCE: The NCCN [National Comprehensive Cancer Network] guidelines try to very strictly adhere to the published data.2 The studies were for doubling times of 10 months or less, and in that situation, they list the preferred drugs, being the 3 FDA-approved agents that we’re talking about, and it does leave open the option for other secondary hormone therapy, like abiraterone. Then with a doubling time of greater than 10 months, they officially say that observation is preferred.
All right, we’re going to go over the 3 completed, published registration studies for the treatment of [patients with] nonmetastatic, castration-resistant prostate cancer [CRPC]. With all these studies, the study designs are largely the same; very, very slight differences.
[In the SPARTAN trial (NCT01946204) for apalutamide] they define nonmetastatic as being imaging by technetium and bone scans and CT scans showing nothing more than pelvic lymphadenopathy less than 2 cm. Anything above the iliac bifurcation was considered to be distant metastasis and therefore excluded. PSA doubling time had to be less than 10 months, and this was a placebo-controlled study with a primary end point of metastasis-free survival [MFS]. This was a novel end point that was really invented by the FDA for this specific scenario; [this was] the first time we ever had FDA approvals in prostate cancer for MFS. Ultimately patients were then followed out through their next line of therapy for PFS2 [second progression-free survival] and followed for overall survival [OS].3
Basic demographics, there is nothing that stands out as being different from the norm. Median age was 74; that’s certainly quite normal, from the 40s up to the 90s. Average patient was 8 years out from their diagnosis. The doubling time was [6 months or less for 70%]. Only a minority of patients were on bone-sparing agents. Roughly 16% of patients had pelvic lymph node disease.
Very positive data. The primary end point—MFS— the apalutamide-treated patients clearly had a much longer MFS than placebo. The median MFS, even from a basis of just staying on ADT, was 16.2 months, so this is not a scenario occurring very quickly. With apalutamide it’s 40.5 months and the hazard ratio [HR] is 0.28, which is just about as good as you can get in a cancer study.4
Importantly, the secondary end point of OS was also positive. The median OS in the placebo arm was on the order of 59.9 months versus 73.9 months in the apalutamide arm. The reason this is critically important is the essential question that comes is everyone’s going to get an antiandrogen sooner or later, so does it really matter if you slow the disease down early and do you lose the benefit later on? The answer definitely is no, the OS is maintained, despite the fact that these placebo-treated patients went on to one of the oral agents later on in their disease course. So earlier is better than later. And the HR is quite strong as well [at 0.78].5
[In terms of the] treatment-emergent adverse events [AEs] [in this trial], there are really no surprises. This is what we expect out of an oral anti-androgen, certainly some fatigue, that was 32.6%, all grades, only 0.9% or so were grade 3/4 with apalutamide. Hypertension is higher in patients on novel anti-androgens than on placebo, and you see a little bit of GI [gastrointestinal] toxicity as well. The falls always pop out on these studies—22.0%, that’s in line with what we’ve seen with other studies, but it’s a real effect and something that we need to pay attention to.
A particular adverse event with apalutamide is rash, so this is something to be aware of, and the first time you use apalutamide, you might be surprised. As many as a quarter of patients will develop a maculopapular erythematous skin rash. It’s generally grade 1/2. I mean, it’s generally not severe, and it happens somewhat later in the course, at a median of 82 days, but often, it’s self-limited and will resolve on its own.
Corticosteroids can be used. It can lead to dose interruptions, certainly, but it’s uncommon that patients have to discontinue it. It just becomes an annoyance that generally can be managed.
[Next], PROSPER [NCT02003924] is the enzalutamide study. Again, a very similar trial design, only slight differences in terms of the pelvic lymph nodes, but same basic study design, followed out for MFS as the primary end point, OS as the secondary.6
The inclusion criteria, again, are very similar. Amazingly similar, really, they’re 74 years old, the PSA doubling time on the order of 4 months at a median.
The results are very much in line, the median MFS in the placebo arm [was 14.7 months], in the same ballpark versus 36.6 months MFS with the use of enzalutamide. So again, very similar results.
The OS data were also very positive, and again, very similar results [56.3 months with placebo versus 67.0 months with enzalutamide], so we’re getting a lot of confidence that we know what happened to these patients in that it portends a survival on the order of 5 years. These are not patients who are going to die of something else; these are the patients who really are going to die of their prostate cancer, and that’s why we want to manage them.7
Paying attention to the toxicities, because this is one of the ways we differentiate between the drugs. The hypertension rate is significant, 43 versus 10 [patients with enzalutamide and placebo, respectively]; the fatigue, 27 versus 3; falls is 12 versus 3. With enzalutamide studies, you’ll always see a certain amount of special attention to some of the CNS toxicities. Patients with a history of seizure were excluded, so they didn’t see that here, but still, you see this profound ischemia and a slightly increased risk of various cardiac outcomes—cardiac failure, myocardial infarction, etc.
The ARAMIS trial [NCT02200614] is the darolutamide study, and again, very similar in design to the others: the ECOG performance status 0 to 1, 1500-patient study, follow-through for MFS with OS as a secondary end point.8
Again, very similar patient demographics across the 3 studies: median age of 74, median doubling time on the order of 4 months, and just like the other studies, only a minority of patients were on bonesparing agents.
Again, we get very similar results, the median MFS on the placebo arm was 18.4 months, but on the treatment arm was 40.4 months [HR, 0.41]. Very strong positive result, and OS data were extremely encouraging, with an HR of 0.69 at a median follow-up of 29 months. These data are not quite as mature; the placebo hasn’t reached median yet.9
Fatigue and asthenia are closer between the treatment arm and the placebo arm than we saw with apalutamide and enzalutamide, which is really one of those CNS toxicities. I don’t know if you’ve seen it in your practice, but you can have a patient on enzalutamide with profound fatigue, stop the drug, and a month later, they feel quite good, despite the fact that you’re not controlling their cancer anymore.
Falls are equal between the arms on this particular study [4.2% vs 4.7%]. Dizziness is equal. Coronary outcomes are fairly close, and the rash is only 2.9% versus 0.9%.

So as we look at the 3 studies, the MFS is similar across these studies; the OS was very similar. Really, all 3 drugs work. They work much the same way. There’s really not a lot of space between them in terms of efficacy, they’re all very similar.
There was a comment earlier about dosing. Remember, apalutamide and enzalutamide are once daily; darolutamide is twice daily.
But the toxicity is where we see a difference between the drugs. Both apalutamide and enzalutamide cross the blood-brain barrier, whereas darolutamide doesn’t. The fatigue, the hypertension are higher with enzalutamide and apalutamide than with darolutamide.
Rash is a special toxicity with apalutamide. The falls are higher in these patients who have the fatigue and asthenia, so probably related to the direct CNS effect of antiandrogens.
[Let’s] open this up for discussion. So what are the practical implications of having both OS and MFS for AR-directed therapy in nonmetastatic CRPC, and does this influence your use of these agents?
VELEZ: I guess we’re using more agents, probably earlier than compared with a few years ago, so practical implications are cost, of course, toxicity for the patients, insurance approvals, and then in some way it affects the sequencing of agents for the future, the type of relapse/ recurrences you can have compared with 5 to 10 years ago. But once you prove the benefit in both MFS and OS, those are the hard end points and I would say that’s a pretty good argument to use them.
GU: I think the OS is really [encouraging] because when you think about sequencing, sometimes you need that OS to convince you. I think it helps us to talk to the patients because everybody has quality of life considerations, but if you can show them these curves, it helps them to understand it’s all relative. Quality of life is bad when you have disease contributing to your poorer quality of life down the road and shorter OS. If you introduce that to the patient, then you can actually have patients who you convert and the hard-core patients who refuse. That’s fine, at least they know the facts, because I think it’s our job as physicians to guide them and I think this enables me to have that good conversation with them.
BRYCE: OS is where it’s at. We need those data. I would have felt uncomfortable with just MFS when we start talking about quality of life. We’re not treating a number. It’s not just about PSA.
REFERENCES
1. Mori K, Mostafaei H, Pradere B, et al. Apalutamide, enzalutamide, and darolutamide for non-metastatic castration-resistant prostate cancer: a systematic review and network meta-analysis. Int J Clin Oncol. 2020;25(11):1892-1900. doi:10.1007/s10147-020-01777-9
2. NCCN. Clinical Practice Guidelines in Oncology. Prostate cancer, version 2.2021. Accessed June 22, 2021. https://bit.ly/3hhH9Or
3. Smith MR, Saad F, Chowdhury S, et al; SPARTAN Investigators. Apalutamide treatment and metastasis-free survival in prostate cancer. N Engl J Med. 2018;378(15):1408-1418. doi:10.1056/NEJMoa1715546
4. Small EJ, Saad F, Chowdhury S, et al. Updated analysis of progression-free survival with first subsequent therapy (PFS2) and safety in the SPARTAN study of apalutamide (APA) in patients (pts) with high-risk nonmetastatic castrationresistant prostate cancer (nmCRPC). J Clin Oncol. 2019;37(suppl 7):144. doi:10.1200/JCO.2019.37.7_suppl.144
5. Small EJ, Saad F, Chowdhury S, et al. Final survival results from SPARTAN, a phase III study of apalutamide (APA) versus placebo (PBO) in patients (pts) with nonmetastatic castration-resistant prostate cancer (nmCRPC). J Clin Oncol. 2020;38(suppl 15):5516. doi:10.1200/JCO.2020.38.15_suppl.5516
6. Hussain M, Fizazi K, Saad F, et al. PROSPER: a phase 3, randomized, double blind, placebo (PBO)-controlled study of enzalutamide (ENZA) in men with nonmetastatic castration-resistant prostate cancer (M0 CRPC). J Clin Oncol. 2018;36(suppl 6):3. doi:10.1200/JCO.2018.36.6_suppl.3
7. Sternberg CN, Fizazi K, Saad F, et al; PROSPER Investigators. Enzalutamide and survival in nonmetastatic, castration-resistant prostate cancer. N Engl J Med. 2020;382(23):2197-2206. doi:10.1056/NEJMoa2003892
8. Fizazi K, Shore N, Tammela TL, et al; ARAMIS Investigators. Darolutamide in nonmetastatic, castration-resistant prostate cancer. N Engl J Med. 2019;380(13):1235-1246. doi:10.1056/NEJMoa1815671
9. Fizazi K, Shore N, Tammela TL, et al; ARAMIS Investigators. Nonmetastatic, castration-resistant prostate cancer and survival with darolutamide. N Engl J Med. 2020;383(11):1040-1049. doi:10.1056/NEJMoa2001342