Roundtable Discussion: Treatment Strategies for Patients With High- Risk Myelofibrosis
Ellen K. Ritchie, MD, discuss the case of a 68-year-old man with high-risk myelofibrosis during a case-based roundtable event.
Ellen K. Ritchie, MD

RITCHIE: Symptom control has gotten a lot of press, so to speak, in academic literature. Would any of you like to make any comments as to what triggers you to initiate therapy for a [patient with] MF?
ROTKOWITZ: It’s a loaded question, right? I think that we will probably talk about pacritinib [Vonjo], momelotinib, and some of the newer agents, which are exciting.
But I think that there is a long [list of] criteria, as per the NCCN [National Comprehensive Cancer Network] guidelines, [to guide that decision], or a modified IPSS.1 I think that [the most valid assessment is to simply] look at the patient and talk to the patient, and I think there is an incredible amount of heterogeneity in this disease.
A lot of patients have overt symptoms that seem a little out of proportion to their level of underlying disease. I don’t feel that I am going to cure any of the patients I see, so it’s a marathon, not a sprint, but I want to [extend the] time to progression to acute myeloid leukemia [AML]. I think that the morbidity associated with chronic cytopenia is real and that 95% of this job is psychiatry in dealing with your patient and getting to know the impact that the symptoms are having on the patient.
I think, historically, I’ve been limited by the therapeutic options. [Options for patients such as this one, with a platelet count] below [50 × 103/L], are incredibly limited, [although the options] are going to change. [The treatment decision is best made] when you see your patient. As much as we say we take care of patients, not numbers, it is a numbers game, but if the patient is symptomatic [due] to cytopenia, that’s a huge trigger for me to do something. Definitely, [if a patient is experiencing] early satiety, left upper quadrant pain, or symptomatic splenomegaly, that is a no-brainer to me. Also, if the cytopenias are [affecting] that patient’s quality of life [in other ways], that’s a trigger for me to intervene.
RITCHIE: I think you are right that it is a marathon, not a sprint. And listening to your patient and trying to figure out the best way to palliate their symptoms is an important aspect of taking care of the patient. Does anyone else want to comment?
FAZAL: I think the goal of the patient is very important. I think that you need to have an up-front discussion with the patient about how much the disease is [affecting] their quality of life, what issues they are experiencing, and what they think the treatment is going to achieve. I think that is important in terms of setting expectations. If a patient is looking to improve their blood counts, the available therapy has certain limitations.
[In comparison, one might have] a patient who has significant symptoms from splenomegaly and from constitutional symptoms. Do we believe that patients whose risk level is intermediate-1 benefit from a JAK inhibitor? There are data that favor that, but the data are not [from randomized studies], so it is a decision that we have to make with each patient.
PROOTHI: It’s a loaded question and has loaded answers. Generally, I go with the symptoms plus the coding system.
[If the risk level is] intermediate to high, that’s a trigger. If the patient has symptoms that affect their quality of life, that’s a trigger.

RITCHIE: As you may know, approximately two-thirds of patients with MF either have anemia or are transfusion dependent.2
PROOTHI: I would say most of my patients are cytopenic vs myeloproliferative.
RITCHIE: How do you approach these patients who are cytopenic? What kind of therapy do you offer?
PROOTHI: [I offer it] only if they need therapy. Not everybody needs therapy. Even if their hemoglobin is 10 g/dL, if they are not symptomatic, I don’t treat them. [In the] same way, if their platelet count is greater than 10 × 103/L, then I don’t treat them, even though they are thrombocytopenic. It all depends on if they need treatment. Because if you transfuse, half of the blood goes into the spleen, so not everybody gets the full benefit of it.
FAZAL: There is [debate] about whether different phenotypes of MF present differently, eg, whether primary MF presents with more cytopenias and secondary MF presents with more proliferative disease. On the other hand, when patients progress, they develop thrombocytopenia as well. I think that thrombocytopenia is seen less often, which is a risk factor that suggests disease progression.
RITCHIE: We don’t have good drugs for patients with cytopenic MF. We don’t have a drug that is necessarily going to improve their anemia and their thrombocytopenia, and that’s frustrating, but it also means that there is a real open space for new drugs and new approaches to this disease.

VARADI: I think the first question that we and the patient have to face [is the goal of treatment]. [Are we looking for] a cure [if we can attempt] a transplant, prolongation of life, or only symptom management? Whether a clinical trial is available depends on where you work. In the communities, it is more difficult. At a teaching hospital or academic center, it is much easier to involve the patients in clinical trials.
RITCHIE: Is it frustrating that it is so difficult to get clinical trials to your patients if you are not close to an academic medical center?
VARADI: Yes. It is a problem because you must make the referral. Sometimes the patient doesn’t want to go; also, you [might] lose the patient. These things are not so simple.
SAMHOURI: For a patient such as this, high risk and aged 68 years, if there is a clinical trial available that we think will benefit the patient, that should always be the first choice. However, for high-risk disease, the question would be if the patient can go to transplant. If the patient can go for a transplant, then the question is, should we give a JAK inhibitor [first]?
Splenomegaly of 30 cm can delay engraftment. We also don’t know if giving a JAK inhibitor before [transplant and] changing the cytokine profile is going to affect graft-vs-host disease. [We also need to consider] how we are going to stop or taper the JAK inhibitor before the transplant so that we don’t [cause] withdrawal syndrome. There are multiple debatable issues here, but the goal should be to go to transplant in a patient such as this, if he qualifies. Before the transplant, giving the JAK inhibitor can improve performance status, especially in a patient who presents with constitutional symptoms.
NATHAN: I don’t feel comfortable giving a JAK inhibitor if the platelet count is less than 50 × 103/L. For this patient, I would certainly investigate a clinical trial or a transplant. I would call the transplant physician and ask if that’s an option.
FAZAL: I think that, as mentioned, the patient would certainly be a candidate for transplant on the basis of his age and high-risk disease. The question is how to optimize patients with constitutional symptoms, splenomegaly, [and] poor performance status. Pacritinib recently received approval for patients who are symptomatic and who have splenomegaly and platelet counts of less than 50 × 103/L, so this patient, who is cytopenic, certainly would be a good candidate to start pacritinib to optimize his condition before evaluating him for transplant.
RITCHIE: In talking to your patients with high-risk MF, who are eligible, and referred for allogeneic [stem cell] transplantation [ASCT], what proportion of your patients receive it? Fifty percent, I think, is a pretty good number, because it is not easy to talk about transplant with your patients. There is sort of a sweet spot, where these patients have to be transplanted before they get too sick to go to transplant, and a lot of patients with primary MF who are high risk, once you have them on a little therapy, may not feel that they are ready to go to transplant. There is a lot of difficulty in getting patients to transplant. Does anybody want to comment on that?
PROOTHI: Is there any age cutoff for the ASCT? Because most of these patients are older.
RITCHIE: I think different transplant centers look at age differently. There are a lot of data to suggest that it is not age but performance status and comorbid illness that contribute to the outcome. So, a 72-year-old, 75-year-old, or even 76-year-old patient who plays tennis every day and has no comorbidities might be a better candidate, for example, than someone who is in their 40s with diabetes, hypertension, and coronary artery disease. It’s difficult to determine exactly who the best candidates are and how big a factor age is in transplantation.
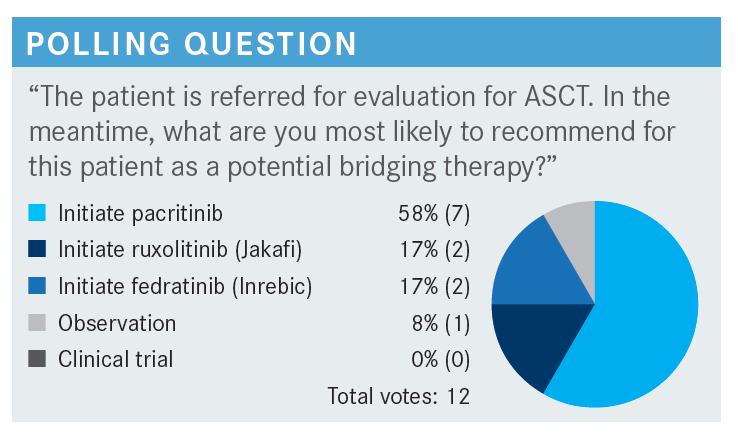
RITCHIE: I find it intriguing that none of you would refer the patient for a clinical trial even though the patient has significant thrombocytopenia, anemia, and a poor prognosis. There are many new, exciting agents and combinations out there for these patients.
ROTKOWITZ: I am in the community at [Sidney Kimmel Cancer Center—Jefferson Health in Philadelphia, Pennsylvania], so I am in southern New Jersey and the Philadelphia suburbs. We have some transplanters who come in from the city to [where we are], a half hour away. They spend a bit of time there, so there is some hybrid [between the] community [and the] city. I also do a lot of [ASCT] and CAR [chimeric antigen receptor] T-cell [therapy]. I chose pacritinib [for a bridging therapy], but I wouldn’t do it without a conversation [with another physician].
I think that there’s a lot of excitement regarding telomerase inhibitors, IL-1 receptor-associated kinase, lysine-specific demethylase 1, phosphatidylinositol 3-kinase, and other agents. There aren’t a lot of good trials that are readily available. I know that at Jefferson they don’t have any first-line trials. Patients are reluctant to cross the bridge to go into the city, or to do so repetitively, so yes, we have patients with AML or [myelodysplastic syndrome] who go [for ASCT].
I wouldn’t decide to start anyone on a therapy without speaking to someone first. We have done a lot of telemedicine, at least for some collaborative assessments for patients. In the past, I was in a community private practice, and then it was quite difficult, because you felt as though you sent your patient out and you never saw them again.
Here, there are multidisciplinary tumor boards. There’s collaboration. [But] I often find that, while I wish it was a 1-way ticket, it turns into a boomerang, and the patient ends up right back with me, and I am stuck with those options. I think that because [MF is] a relatively rare disease, there isn’t a tremendous amount of clinical trials.
RITCHIE: I think that there’s some truth to that, and there’s a difficulty, also, in the eligibility for some of these clinical trials. The FDA has mandated a higher platelet count than you see in most patients who present with cytopenias and MF. I [also] think that COVID-19 has made some changes in the way that clinical trials are conducted. At my center, a patient must come in for the initial evaluation, potentially an MRI and a bone marrow biopsy, [to obtain] first medication.
[Beyond this,] I often treat most of [the patients] with a community doctor and with telemedicine in between. So I think that [if there’s a silver lining from the COVID-19] pandemic, it is that it gave us this ability, the need, to use telemedicine and maybe conduct clinical trials better. I think that is probably true of stem cell transplant as well. Are any of you in a more rural area where you find it difficult to connect with a transplant team?
SILVA: Yes. I am approximately 30 minutes north of Pittsburgh, Pennsylvania. Normally, in a case such as this, I discuss with the transplant service ahead of time whether they would [prefer] to see the patient initially or to see the patient later. The problem is that patients in the area are resistant to travel, and travel sometimes just means crossing a river. Unless the patient is young and motivated, they just don’t want to go to Pittsburgh.
RITCHIE: It sounds to me as though most of you believe the major obstacle to stem cell transplant or clinical trial is that patients just don’t want to go. Is that what most of you think?
SILVA: This is a very regional thing about Pittsburgh. I’m not sure [about] other parts of the country. I was in Philadelphia for the first part of my training, and I didn’t notice this [phenomenon when I was there]. But I think that in Pittsburgh patients are tied down geographically and don’t want to move around.

PROOTHI: Before, there were not too many choices. Now, we have a little more choice. Before we just had ruxolitinib [Jakafi], and then the fedratinib [Inrebic] came along, and now we have [study results showing] that we can use it even in the first line with a low platelet count. [From the Data].4
In our practice, we have a transplanter come to our facility twice a month, so that makes it easy, and as you said, telemedicine has made a difference. When [the transplanter] comes to the area, he comes to see the patients. In the meantime, he generally tells us what he thinks we should do.
If a patient has low risk, we don’t do much. If the risk status is intermediate or high, [we determine] if the patient is eligible for transplant on the basis of performance status and organ functions. And if [the patient is eligible], then generally the [transplanter] tells us what we should be doing [for the patient] in the meantime.
RITCHIE: So you and the transplanter decide [together] what the bridging therapy is going to be?
PROOTHI: Right.
RITCHIE: Does anyone want to comment on [which patients] you believe are ineligible for transplant, with respect to factors besides age?
MEWAWALLA: I think patients with poor performance status [are ineligible]. I just think that patients with MF have a very poor performance status. They tend to do poorly [and have] massive splenomegaly. Those patients just don’t do well with transplant.
VARADI: Or patients with poor kidney function or heart failure.
RITCHIE: So, [you’re saying] comorbid illness, which has been proven in the transplant literature.

FAZAL: I think [that for] patients who have symptomatic splenomegaly and constitutional symptoms with thrombocytopenia, you could consider using [pacritinib] up front. In terms of the patients who are on ruxolitinib and are severely thrombocytopenic even on a lower dose, [and] if they have symptomatic splenomegaly and constitutional symptoms, you could consider switching the patients from ruxolitinib [to pacritinib].
RITCHIE: How would you counsel a patient receiving pacritinib? What would your advice be? What would you want to discuss with that patient before starting?
FAZAL: I think the [gastrointestinal symptoms are] the major thing that I would talk to the patient about. I guess, of course, there was a clinical hold that was related to cardiac events. I would discuss [this] with the patient just in case they [were to] look online [and see that]. I would [explain] that no major concern was seen on subsequent data analysis.
RITCHIE: The first patient that I put on pacritinib as a commercial drug saw a dentist on the same day that she was starting the pacritinib. The dentist put her on clarithromycin, and she developed [severe] diarrhea, and it was hard to know what the cause was. [In the end], it looked as though it was the clarithromycin. One of the difficult things about treating these patients is being in control of what other medications the patient is receiving when [they are being treated by] other doctors [at the same time]. [This is an example of one] of the difficulties of starting therapy in patients such as this.

PROOTHI: If we are starting [therapy with pacritinib] in a patient who has a platelet count of less than
50 × 103/L, and the platelet count drops, where is the cutoff? Do you stop [treatment when the platelet count reaches] 25 × 103/L? Or do you transfuse the platelets and keep [using] the drug?
RITCHIE: On our trial of pacritinib [NCT01423851], we usually kept [treating] these patients.5 We didn’t stop the drug unless the patient wanted to stop the drug. It took a while before the patients saw any improvements in their blood counts, so I think that some patients did receive transfusions of platelets while initially on therapy.
PROOTHI: The burden of symptoms and the transfusion dependency [would most influence my decision].
RITCHIE: I certainly think that blood counts and symptoms are very important in deciding whether to start a palliative therapy. JAK inhibitors are not curative; they are just palliative. Are there any comments on that?
FAZAL: The only comment I would make is that the only curative option we have is transplant, and when these patients are transplant eligible, you do see better status prior to transplant, of course. Using JAK inhibitors certainly has some role in that setting to optimize a patient’s condition, probably leading to better outcome after transplant.
RITCHIE: I think that your goal for the patient is also important. For a patient with a big spleen who is going to transplant, [the goal] becomes an important [consideration] in the decision to use a JAK inhibitor prior to transplant to decrease spleen size. The problem with [including] risk, age, and frailty [in this list of influential factors] is that none of these [drugs] are curative [and should only be used] if you feel that the therapy would improve the patient’s quality of life.
I have a patient right now who has amyotrophic lateral sclerosis and who is slowly deteriorating from that disease, but he also has MF, and his spleen and liver began to grow. Even though his performance status is poor, it was reasonable to start a JAK inhibitor just to make him more comfortable. So there are [many different] things we look at to decide whether to treat a patient and which JAK inhibitor to use.
REFERENCES:
1. NCCN. Clinical Practice Guidelines in Oncology. Myeloproliferative neoplasms, version 3.2022. Accessed August 30, 2022. https://bit.ly/2E77tIB
2. Tefferi A, Lasho TL, Jimma T, et al. One thousand patients with primary myelofibrosis: the Mayo Clinic experience.Mayo Clin Proc. 2012;87(1):25-33. doi:10.1016/j.mayocp.2011.11.001
3. FDA approves drug for adults with rare form of bone marrow disorder. FDA. March 1, 2022. Accessed August 31, 2022. https://bit.ly/3wU4M8q
4. Harrison CN, Schaap N, Vannucchi AM, et al. Safety and efficacy of fedratinib, a selective oral inhibitor of Janus kinase-2 (JAK2), in patients with myelofibrosis and low pretreatment platelet counts.Br J Haematol. 2022;198(2):317-327. doi:10.1111/bjh.18207
5.Verstovsek S, TalpazM, Ritchie E, et al. A phase I, open-label, dose-escalation, multicenter study of the JAK2 inhibitor NS-018 in patients with myelofibrosis. Leukemia. 2017 Feb;31(2):393-402. doi:10.1038/leu.2016.215

Survivorship Care Promotes Evidence-Based Approaches for Quality of Life and Beyond
March 21st 2025Frank J. Penedo, PhD, explains the challenges of survivorship care for patients with cancer and how he implements programs to support patients’ emotional, physical, and practical needs.
Read More