Overcoming Barriers to Genomic Testing in Community Care Settings
Genomic testing has grown in importance as the number of targeted anticancer therapies rises. Early genomic testing can help to guide treatment choices.
Robert L. Coleman, MD

S. Michelle Shiller, DO, MSPT

As targeted anticancer therapies proliferate, companion diagnostics have become an equally indispensable part of the clinician’s tool box. This effective combination helps determine the genetic makeup and protein expression status of a patient's tumor, while informing the oncologist's treatment choices.
Genomic testing appropriate to the type and stage of tumor is the way forward to ensuring that patients receive maximum benefits from targeted therapies. This increased focus on the use of biomarker and genomic testing to guide treatment for patients with cancer has found wide acceptance in comprehensive cancer centers and large research-based institutions. But by all accounts, testing uptake still lags among community-based oncology practices, although the situation does appear to be improving.
Robert L. Coleman, MD, chief scientific officer at US Oncology Research, points out that drug development is what is driving genomic testing. As new targeted therapies become the standard of care, testing rates are increasing, albeit more slowly than he would like.
“For many tumors, whether or not you test depends on what you can do with the information. Look at how PARP inhibitors have affected my field of ovarian cancer: The only approval for a PARP inhibitor we had in 2014 was for patients with germline BRCA-positive recurrent ovarian cancer,” he says. “Then we studied [and got approval] as a third-line therapy, and then as a maintenance therapy for recurrence, and then as a maintenance therapy for primary. As we’ve gotten approval as a frontline indication, we now need to know the BRCA mutation status at the time of diagnosis, and clinicians do need to adapt to that.”
Where We Are Now
S. Michelle Shiller, DO, MSPT, a molecular geneticpathologist and associate director of genomic and molecular pathology services for PathGroup/PBM in Dallas, Texas, says that message seems to be getting through to clinicians. “Since 4 out of every 5 patients with ovarian cancer present with advanced disease, they should get broad genomic profiling instead of testing just for BRCA1 and BRCA2 mutations,” she says. “Most orders we see are indeed for the broad panel, so that’s a good development.”
In general, Shiller finds that clinicians are getting better about ordering testing. “There’s still a gap between what the guidelines or FDA approvals say and what we actually see in the field,” she says. “Too often, a clinician doesn’t test until after a patient has progressed or escaped a prior therapy.”
Testing for non–small cell lung cancers (NSCLCs) has been a relative bright spot. “Testing uptake has been the best with NSCLCs but we’re still not seeing enough,” Shiller says. “All advanced NSCLCs should receive broad genomic profiling at diagnosis, and all early-stage lung cancers should get screened for the presence or absence of an EGFRmutation at diagnosis. We’re not seeing enough early-stage testing yet, not even in lung cancer.”
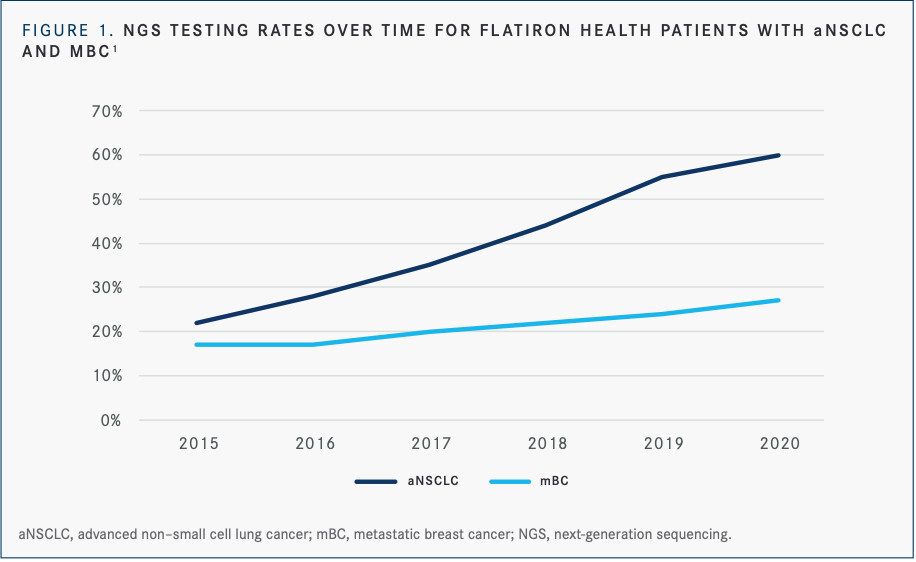
An analysis of testing patterns with next-generation sequencing (NGS) for patients with advanced NSCLC (n = 29,572) as well as for patients with metastatic breast cancer (n = 12,175) from January 2015 to May 2020 showed significant improvements in NGS testing rates for NSCLC over 5 years (22% in 2015 vs 60% in 2020) and more moderate improvements in testing rates for breast cancer (17% in 2015 vs 27% in 2020) (FIGURE 1).1
Shiller also notes that most—but not all—institutions are ordering mismatch repair testing by immunohistochemistry on all colon and endometrial cancers. “This gap needs to be closed, because aside from screening for the potential to respond to an immune-based therapy, it also screens a patient for hereditary predisposition. For colon, only about 75% to 80% of all-comers [have those orders], either in diagnostic biopsy or a resection,” she says. “In endometrial [cancer], it’s a bit lower at about 70%. But these are part of the NCCN [National Comprehensive Cancer Net-work] guidelines and there’s a lot of data to drive this practice, so testing rates should be at 100%.”
Possible Barriers to TestingTrends in real-world genomic testing have become a subject of widespread interest in oncology. A glance at the programs from the 2021 American Society for Clinical Oncology (ASCO) Annual Meeting and the 2021 ASCO Quality Care Symposium hints at both insufficient community-based genomic testing and optimism that testing is on the upswing. Dozens of ASCO abstracts this year focused on some aspect of documenting—and then improving—genomic testing at the community-practice level.
Some commonly perceived barriers to full testing include:
- testing prices and reimbursement;
- racial and socioeconomic disparities;
- tissue requirements; and
- turnaround times.
Here’s a closer look at some questions these studies are asking and attempting to answer.
Testing Prices and Reimbursement
For years, technology development has followed a familiar pattern: initial to-market iterations are prohibitively expensive and relatively limited in performance. Then, within a few years, price drop dramatically even as features increase.
Genomic testing is no different. Everyone agrees that genomic testing prices have decreased even as they have become able to offer testing panels of 450 to 500 genes. In Coleman’s experience, genomic testing can range from about $250 to $2500 per testing panel. “It’s clear that pricing is on a downward trend, and, of course, the actual cost of testing varies from what is charged,” he says. “Reimbursement trends are improving because drug payment approvals are tied into knowing [a patient’s genetic status].”
Tracking genomic testing pricing in detail has been challenging because of all the variables involved. At ASCO 2021, one Merck-funded study set out to describe historic trends in the average allowed cost for NGS testing.2
The retrospective database study looked at 4 years of patient claims for tumor and/or germline sequencing tests. The authors, led by Merck’s Kaushal Desai, PhD, found that average allowed amounts for NGS tests varied from $1269 to $2058 per test during the study period, which concluded in 2019. They found even greater variability in subgroup analyses that included the patients’ region, age, gender, and cancer type. For hereditary cancers, the average allowed amounts varied from $1722 to $2249 per test. Pricing and utilization were more consistent over time, with prices gradually decreasing.
Tests that focused on the newer biomarkertumor mutation burden showed an even larger range: $438 to $3700 each.
“Currently, coverage and reimbursement of NGS tests is one of the key policy challenges that needs to be addressed,” wrote Desai et al. “Hence, development and adoption of policies is needed to ensure efficient integration of NGS testing into the US healthcare system.”
Figure 1
Racial and Socioeconomic Disparities
Teasing out the ways in which health disparities interact with genomic testing trends in community oncology is even more challenging than analyzing pricing trends. And yet, for Coleman, the issue has a certain simplicity: “If NGS testing is tied to insurance coverage, there are going to be disparities because there’s a massive amount of disparity built into insurance coverage in the United States,” he says. “Anecdotally, I’ve found that patients who have Medicare can usually get testing covered.”
One study at ASCO 2021 that garnered attention was a retrospective observational study led by Debora Bruno, MD, of University Hos-pitals in Cleveland, Ohio, that investigateddifferences in Black vs White race among patients with NSCLC undergoing biomarker testing and clinical trial enrollment. Conducted using the Flatiron Health database, the study found that comparable numbers of White patients (76.4% of 9800) and Black patients (73.6% of 1300) underwent at least 1 single molecular test or comprehensive genomic analysis (P = . 0 3).3
Bruno et al found that the use of NGS-based testing, which is recommended by the NCCN Clinical Guidelines in Oncology for patients with advanced/metastatic NSCLC,4 reflects the most notable disparity between Black and White patients. NGS was performed among 50.1% of White patients and only 39.8% of Black patients (P < .0001).3 “Black patients had more than a 10 percentage-point difference in receiving this testing vs White counterparts,” they concluded. “Also, Blacks were 55% less likely to be treated within a clinical trial...as many second line and beyond trials utilize molecular targets as inclusion criteria.”
An ASCO 2021 poster from the Association of Community Cancer Centers (ACCC) and the LUNGevity Foundation used a mixed-methods approach to identify barriers to equitable precision medicine access among underserved patients with NSCLC. Using a national survey of clinicians and one of patients, the authors triangulated survey data with data from clinician and patient focus groups to probe issues of concern.5
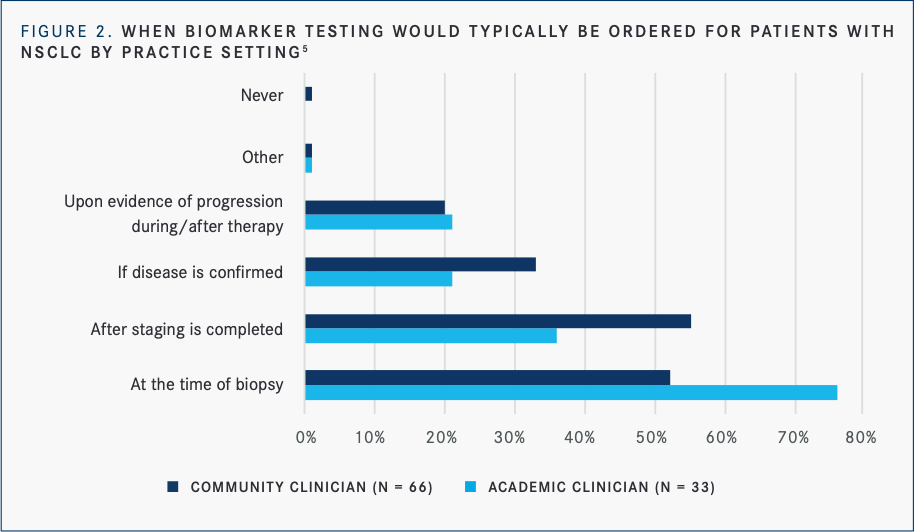
A total of 78% of clinicians said they were “very” or “extremely” likely to discuss biomarker testing with patients; less than 7% of clinicians said they would not discuss testing with their patients at any point during the management of their disease. Further, only half of community clinicians ordered testing at the time of initial biopsy, while nearly three-quarters of academic clinicians did. Eighty-five percent of LUNGevity-connected patients received biomarker testing, yet only about half of the underserved population did (FIGURE 2).
“Notably, more than a quarter (27%) of underserved patients who have undergone biomarker testing do not know their results,” wrote the authors, led by Leigh Boehmer, PharmD, BCOP, chief medical officer of the ACCC.
There were also differences observed in the study for the likelihood of ordering biomarker testing for a patient with NSCLC based on socioeconomic status and health literacy. “[We] identified disparities in offering biomarker testing and results to patients with known or presumed low socioeconomic status [SES] and/or health literacy. This was supported by patient survey data, which showed biomarker testing was proactively offered to only 40% of low-SES patients,” Boehmer et al wrote.
Clinicians surveyed responded that they were extremely (57%) likely to order testing for a high SES patient, compared with 32% for low SES or homeless patients. Sixty-three percent of clinicians were extremely likely to order testing for a patient with a high perceived health literacy, compared with 32% for patients with a low perceived health literacy.
Tissue Requirements
As for genomic testing, tumor tissue is a finite resource. Shiller believes running a broad sequencing panel is the best way to make use of the limited tissue obtained in a biopsy. “It’s better tissue stewardship to choose the broad profiling because you have less likelihood of running out of tissue before you answer the clinical question,” she says. “Some doctors start by ordering individual genes, but then after 3 or 4 additions to the order, you’ve paid for the NGS that would actually tell you more and you may have run out of tissue to boot.”
However, tissue requirements may become less of an issue in the future as liquid biopsy tests can gather ever-increasing amounts of data from a simple blood draw.
Turnaround Times
According to Shiller, an industry standard for turnaround time is 10 days from when the specimen arrives at the lab.
In Coleman’s opinion, genomic sequencing turnaround times can be a major imped-iment for starting patients on targeted therapies. “That’s particularly true if the patients are too ill to wait,” he says. “That often leads us to do a more general treatment like chemotherapy, so the turnaround time is key for increasing testing uptake in the community. Liquid-based assays currently have a leg up in timing, but it would be great to reduce turnaround even more.”
ASCO recently funded a study that probed the issue of turnaround time in managing patients’ treatment. An ASCO task force, including representatives from the American Cancer Society National Lung Cancer Roundtable and patient advocates, conducteda study to assess opinions on biomarker testing and treatment practices for patients with advanced NSCLC among US oncologists. Nearly 2400 ASCO members who treat at least 1 patient with NSCLC per month, including lung cancer specialists and general oncologists (59% were academic physicians and 41% were community based), received a survey.6
Nearly all respondents (98%) believed they should receive biomarker results within 1 or 2 weeks of ordering, yet 37% waited an average of 3 or 4 weeks for results. About a third of respondents who usually wait 3 or 4 weeks said they initiate a non-targeted systemic treatment while waiting. Respondents from community practices were more likely to initiate non-targeted, systemic treatment if results were not available after 2 weeks (59%, compared with 40% of academic respondents). Additionally, oncologists with fewer years since training were more likely to express concerns and delay treatment while waiting for biomarker testing results.
“Respondents indicated that treatment decisions are impacted by delays in bio-marker test results,” wrote the authors, led by Kathryn Finch Mileham, MD, chief of the Section of Thoracic Medical Oncology at Levine Cancer Institute/Atrium Health, Charlotte, NC. “Clinicians should be informed about when it is safe and appropriate to defer treatment while biomarker testing is pending. Respondents suggest that diagnostic biomarker testing companies should strive to expedite results.”
Comparatively in the LUNGevity survey, the turnaround process for receiving testing results took over 2 weeks for 23% of community clinicians vs for 3% of academic clinicians.5
MYLUNG Consortium Leads the Way
In order to fully understand the scope of genomic testing in community settings, large organizations are conducting research projects to document current trends in testing.
Educational programs and policies are being created to move toward better testing uptake, including to ensure that all relevant genes are tested for across tumor types and to stay up to date with the latest recommendations.
The US Oncology Network MYLUNG consortium is a step in that direction, according to Coleman, the program’s principal investigator. “We want to understand what’s happening now and eventually push our clinicians to improve upon current testing rates,” he says.
MYLUNG is an ambitious 3-protocol study that ultimately will include some 12,000 patients with NSCLC in the US Oncology Network over a 5-year period. The first results of protocol 1 were presented at ASCO 2021.7
This retrospective, observational chart review included nearly 3500 patients with metastatic NSCLC who started first-line therapy between 2018 and early 2020. Investigators led by NicholasJ. Robert, VP of Medical Affairs for Ontada, assessed testing rates for ALK, BRAF, EGFR, ROS1, and PD-L1; use of full NGS panels; time from diagnosis to therapy initiation; turnaround time from biomarker orders to results; and time from diagnosis to test results.
Robert et al found that 90% of patients had at least 1 biomarker tested, but only 46% had all 5 tests. NGS testing increased from one-third to 45% over the time of the analysis (P < .0001). The median time to starting therapy was 35 days (range, 22-55) and median turnaround time from orders to results received ranged from 10 to 15 days for individual biomarkers. The median time from diagnosis to biomarker results ranged from 13 to 21 days by biomarker.
Guidelines for genomic testing in NSCLC have changed since 2018, however, based on now available targeted therapies for RET, MET, KRAS, and other alterations. “When we wrote the initial MYLUNG protocol, we focused on just the metastatic recurrent incurable patients, but while we were writing the protocols, additional drugs became available and approved in the frontline setting,” Coleman says. “We realized the study and protocols needed to evolve to reflect the temporal changes happening in practice. We’ll be looking at curable patients who are getting testing for adjuvant therapy.”
Protocol 2 will be a prospective observational trial at 11 US Oncology sites that will enroll 1000 patients. “We will monitor the real-world patient journey from presentation through their first line of cancer therapy,” says Coleman. “This will help to understand the types of patients presenting and the barriers to molecular testing across different types of practices with varying levels of expertise, not only in lung cancer but also in molecular diagnostics.”
Protocol 3, to conclude in 2026, will be a series of prospective trials in 20 practices that will look at ways to improve upon the data gathered in the earlier protocols. “Eleven of these sites will be serving as their own controls for the introduction of new standard operating procedures or new testing methodologies,” Coleman notes.
Use Pathologists as a Resource
But how can community oncologists who are not part of larger organizations ensure that they remain up to date in their genomic testing protocols? Shiller advocates forging closer ties between pathologists and clinicians to increase genomic testing to appropriate levels.
“As pathologists, we make that final diagnosis. There’s a clinical impression, there’s a radiologic impression, but we seal the deal. So we’re the key to helping clinicians get comfortable with newer testing protocols,” she says. “I would tell clinicians to find a lab that will partner with them and make themselves readily available for discussion.”
Alternatively, she says, community oncologists can ask their current pathologists to make the connection. “Call them and say, ‘Hey, I want you to own genomic testing. Please do the research and let us know what you recommend,’” she advises.“I also recommend working with your pathologists to set up standard delegated orders for genomic testing. That helps ease the burden on clinicians while ensuring that patients receive optimal care.”
Both Shiller and Coleman see participation in a molecular tumor board as an important educational opportunity for community clinicians. “PathGroup runs a few molecular tumor boards with different institutions throughout each month,” Shiller says. “We’re seeing clinician interest go through the roof, which I think is hugely positive, especially since we’ve seen a fairly significant increase in testing since we started the tumor boards. We’re just trying to get everyone talking about what’s appropriate in genomic testing.”
REFERENCES
1. Vanderwalde A, Ma E, Yu E, et al. NGS testing patterns in advanced non-small cell lung cancer (aNSCLC) and metastat-ic breast cancer (mBC): OneOncology (OO) sites compared to Flatiron Health Nationwide (NAT). J Clin Oncol. 2021;39(suppl 28):288. doi:10.1200/JCO.2021.39.28_suppl.288
2. Desai K, Hooker G, Gilbert K, et al. Real-world trends in costs of next generation sequencing (NGS) testing in U.S. set-ting. J Clin Oncol. 2021;39(suppl 15):e18824. doi:10.1200/JCO.2021.39.15_suppl.e18824
3. Bruno DS, Hess LM, Li X, et al. Racial disparities in biomarker testing and clinical trial enrollment in non-small cell lung cancer (NSCLC). J Clin Oncol. 2021;39(suppl 15):9005. doi:10.1200/JCO.2021.39.15_suppl.9005
4. NCCN Clinical Practice Guidelines in Oncology. Non-small cell lung cancer, version 6.2021. September 30, 2021. Accessed October 15, 2021. https://www.nccn.org/professionals/physi-cian_gls/pdf/nscl.pdf
5. Boehmer L, Roy UKB, Schrag J, et al. Identifying barriers to equitable biomarker testing in underserved patients with NS-CLC: A mixed-methods study to inform quality improvement op-portunities. J Clin Oncol. 2021;39(suppl 28):123. doi:10.1200/JCO.2020.39.28_suppl.123
6. Mileham KF, Roy UKB, Bruinooge SS, et al. Physician concern about delaying lung cancer treatment while awaiting biomarker testing: Results of a survey of U.S. oncologists. J Clin Oncol. 2021;39(suppl 15):9067. doi:10.1200/JCO.2021.39.15_sup-pl.9067
7. Robert NJ, Nwokeji ED, Espirito JL, et al. Biomarker tissue journey among patients (pts) with untreated metastatic non-small cell lung cancer (mNSCLC) in the U.S. Oncology Network community practices. J Clin Oncol. 2021;39(suppl 15):9004. doi:10.1200/JCO.2021.39.15_suppl.9004