Roundtable Discussion: Raz and Participants Debate When and How to Use Osimertinib Based on Available Data
During a Targeted OncologyTM Case-Based Roundtable event, Dan J. Raz, MD, MAS, and peers discussed the challenges of testing in lung cancer and treatment.
Dan J. Raz, MD, MAS


During a Targeted OncologyTM Case-Based Roundtable event, Dan J. Raz, MD, MAS, and peers discussed the challenges of testing in lung cancer and treatment.
RAZ: Are any of you experiencing barriers to treatment, such as having insufficient tissue or testing taking a long time? Do you have an in-house test, and where are you sending it?
MARLA: We don’t have any particular challenges when we send them out for testing. In my case, I can say there are no specific challenges, but typically our oncologists order the tests because the patients all have an established oncologist that I would let order the testing.
PAUL: We typically have an automatic system. Anybody who gets resected and has an adenocarcinoma automatically gets EGFR, ROS1, and PD-L1 testing. Until recently, it was useless because there were no actionable data to use any of it, but recently things have changed. But we never got any backlash. I think the bigger challenge is we were sending each of those tests to 3 different laboratories to get an answer for our specimens, and it’s not very efficient, it takes longer, and it’s probably better to send it to FoundationOne or some other large lab or, if you’re in an academic center, to do it yourself. I think the next real challenge is making sure that any of these specimens get sent and done together.
RAZ: You’re doing a more limited panel. If those tests are negative, are they sending the sample for NGS [next-generation sequencing], or is that just in the metastatic setting?
PAUL: They haven’t really done it for the early-stage lung cancer tests— sending for NGS—but there are some other indications that you potentially could act on. It’s in the stage IV or advanced setting that they’re doing that. My proposal is that if you’re going to do it, you might as well just do NGS for the whole specimen and then you’re done. One of the barriers is that sometimes insurance companies don’t want to pay for it with no indication for it.
RAZ: In my hospital we have an in-house test, and these have a second test that is in-house, but it’s complicated, and it takes a very long time to get results back. However, I have noticed less of an insurance issue just recently in the last year or so. I don’t know if other people have experienced that, but it seems like a lot of insurance companies are just covering everything now. Is this pretty much just oncologists ordering the test, or does anyone here order the test when the pathology comes back?
MARLA: For us, it’s automatic, so it was initiated by our group to do that and is automatically done.
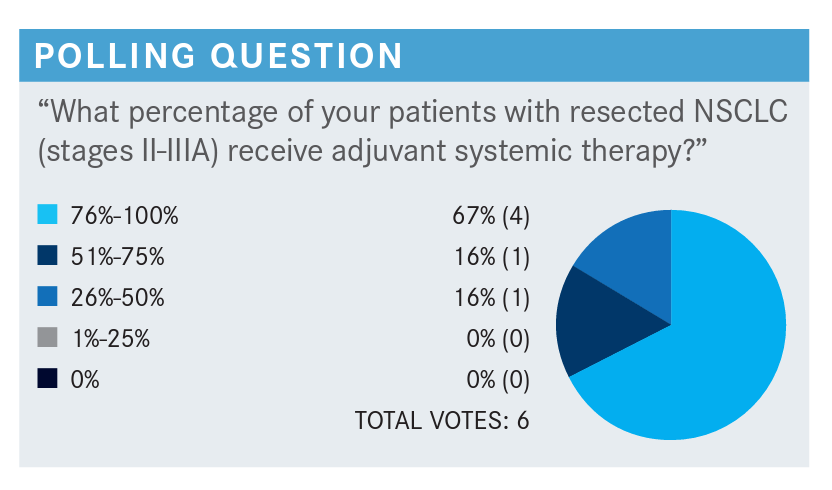
PILUIKO: Same thing for us. It’s an automatic test but usually the oncologist pays attention for everything, so we don’t have many issues in getting the testing done.
RAZ: For patients like this with large tumors, are any of you giving neoadjuvant therapy here? In my institutions, we have several neoadjuvant trials, but are any of you giving patients with a clinically N0 tumor that is on the large side neoadjuvant therapy?
PAUL: We do it in select cases. If someone has a large tumor, they’re fairly young, and they have a good performance status, then we will consider doing it.
MARLA: In our institution, mostly no, especially with N0.
RAZ: Are most of your patients seeing an oncologist before they see you, or how does that work? Or are you sending patients to an oncologist before if they haven’t seen one?
MARLA: Typically, they always see an oncologist even if it’s a straightforward stage I cancer diagnosis. Sometimes, if they can’t fit in and if they are ready to go, we go straight to operating and then they go to see [the oncologist]. Typically, we try to have them establish their stage first with an oncologist.
PILUIKO: I would say at least 70% of my patients already have an oncologist on board, and if they haven’t seen them before they see me, they will be seeing them before surgery.
ONUGHA: Yes. Mostly our patients see the oncologist before surgery unless they present with advanced disease. Most of the time they get referred to us, and then we see them and usually we plan surgery. Once we get the pathology results, we send them to the oncologist.
PILUIKO: Very impressive data—I have a question, however. How do you explain that there’s such a dramatic difference in disease-free survival and the overall survival [OS] is so poor?
RAZ: I think part of the issue is if you look at the number at risk, as you get out to 3 years, there is a very dwindling number of patients because we don’t have that. This study was also stopped early because there was such an impressive disease-free survival change, and that was the primary outcome.
They started off with 233 and 237 patients in [the osimertinib and placebo] arms, respectively, and by 3 years, we are down to 39 and 33 patients; by 4 years, there’s 1 patient left. So, I don’t think those are necessarily deaths, but there have not been a lot of patients who have been on this study for more than 3 years. I think there’s a big question mark long term, what these curves are going to look like.
PAUL: I think it’s a statistical thing. You don’t have enough events that you need to differentiate.
RAZ: I agree with you. On top of that, the survival is really good in both groups.
PAUL: I think one of my problems with this study other than the OS is it’s only in patients who underwent a lobectomy or greater. So it doesn’t have patients who have had sublobar resections, wedge, or segmentectomy. I don’t think they, at least in the journal paper, have anything on the quality of the surgical operations— how many lymph nodes were taken, how many stations were biopsied. I think there are some of those data but not all of them.
RAZ: [Lobectomy or greater] makes sense because these are patients with IB and higher disease. So probably not the type of patients that you would, generally speaking, do sublobar resections for. But I do think it’s an interesting question: How does this therapy change things in patients who don’t have the best pulmonary function? Can you [do] more now that we have this medication?
Any other concerns about the OS? [Will you wait to use this, or will you give osimertinib to all eligible patients?]
SCHWARTZ: I think that’s going to be up to the oncologist where I work. We’re going to refer to them and consider it, but we’re not going to be the ones pulling the trigger on giving them this medication.
MARLA: A lot of it depends on where you are working, based on your oncologist. But at my place, I think they would definitely plan to use it in all eligible patients. At least most of the oncologists who are seeing patients with lung cancer [would].
RAZ: Generally speaking, if you go by the book for stage II and IIIA patients, they should get cytotoxic chemotherapy and then adjuvant osimertinib for 3 years after that. Have any of you had the experience of treating patients like this where perhaps the oncologist skipped the chemotherapy and went right to osimertinib? Or have some of you been seeing, for those of you who have had patients who received adjuvant osimertinib, patients getting chemotherapy [first]?
ABRAHAM: Most of our oncologists are pretty conservative…so they give them standard cytotoxic chemotherapy and try to give it afterward in some other circumstances. I think a lot of them are looking for the OS data before they will do anything, and I think the last thing is it depends where you go. You go to some places where they are not quite following National Comprehensive Cancer Network guidelines: “This stuff is less cytotoxic, so I am just going to give it to all of my patients. Why not?”
PILUIKO: I think our oncologists are very aggressive with that, so I think everyone who is eligible should probably get it.
RAZ: We ask that question about chemotherapy first. I spoke with one of the oncologists who sits with me in clinic and asked her about that and she said, “No, we have to go by the book,” and then I referred her a patient who turned out to have EGFR-mutated disease who had stage II lung cancer, and I saw the patient back a few months later, and she got osimertinib without the chemotherapy. I asked the oncologist and I said, “I thought you were going to give the chemotherapy first,” and she said she was a healthy woman and really didn’t want chemotherapy. So it will be interesting to see in real practice how this is delivered.
What about—I know again this is the oncologist ordering—but have you heard of any insurance issues, for example? This is a very expensive medication; oftentimes these medications have high co-pays. Have any of you heard complaints from the patients about the cost?
ABRAHAM: Yes. We definitely encountered that as declared in our tumor board with regard to cost and being prohibited for patients to get on them.
MARLA: I hear this occasionally. Most of the time they don’t mention cost on the tumor boards.
PAUL: I think it’s pretty new to use in this setting too, so I don’t think people have a lot of experience.
RAZ: Is anyone working in underserved communities where perhaps insurance plans are much more restrictive?
PAUL: Our health system covers an intercity hospital in Newark, New Jersey, where a lot of people have either some form of Medicaid or no insurance, and the oncologists try to get the drug companies to pay for it pro bono or their state to cover it, but a lot of times it’s limited. You’re not getting the state of the art, unfortunately.

RAZ: What about for patients with stage IB, given the lack of OS data? A lot of you made comments about the oncologists making the decision, but what is your opinion? What is your enthusiasm or your oncologist’s enthusiasm for using [osimertinib] in IB disease?
PAUL: I think it depends on waiting for the OS data. I think in some cases they’ll get it. The oncology group I work for said they will give it to patients if they are at a high risk for recurrence like tumor size or some bad features of the tumor. They will give them cytotoxic chemotherapy followed by this if needed.
RAZ: Are you presenting all of your patients at tumor board or selectively presenting patients at tumor board? I know it’s different at each institution.
ONUGHA: Yes. We selectively present patients at tumor board.
MARLA: We present all of the cases, even if it is straightforward stage I.
REFERENCE
1. Wu YL, Tsuboi M, He J, et al; ADAURA Investigators. Osimertinib in resected EGFRmutated non-small-cell lung cancer. N Engl J Med. 2020;383(18):1711-1723. doi:10.1056/NEJMoa2027071
