Roundtable Roundup: Surveying Oncologists on Endometrial Cancer Treatment
Bhavana Pothuri, MD, and Krishnansu S. Tewari, MD, discussed the treatment options of a patient with advanced endometrial cancer and participants chose an immune checkpoint inhibitor to use for therapy.
In separate virtual live events, Bhavana Pothuri, MD, and Krishnansu S. Tewari, MD, discussed the treatment options of a patient with advanced endometrial cancer and participants chose an immune checkpoint inhibitor to use for therapy.
CASE SUMMARY
In August 2021, a 64-year-old postmenopausal woman presented to her gynecologist with abnormal uterine bleeding lasting 4 months. She underwent menopause at 55 years of age. She is a widow, has no children, and lives alone. She has a medical history of gravida 1, para 0; arthritis; obesity (body mass index = 40); and hypertension well controlled with once-daily angiotensin-converting enzyme inhibition. Her 2020 Papanicolaou test was negative, and ECOG performance status was 1.
She was counseled on surgical options, then scheduled accordingly. She had stage IA, grade 1 endometrial cancer. Immunohistochemistry/molecular testing results showed a mismatch repair deficiency and microsatellite instability–high status, and 3+ estrogen receptor–positive disease.
In August 2022, she reported intermittent pelvic pain over prior 4 weeks. CT scan of the chest, abdomen, and pelvis (CAP) suggested relapsed/metastatic disease with involvement of 1 right external iliac lymph node. Carboplatin/paclitaxel (6 cycles every 4 weeks) was administered, chemotherapy was well tolerated, and a complete response was recorded at end of regimen.
In April 2023, disease relapse was documented on routine follow-up. CT CAP scan showed heterogeneously enhancing mass in right suprarenal space, multiple bilateral pulmonary nodules, and a new right internal iliac lymph node (in addition to the previously observed positive lymph node).
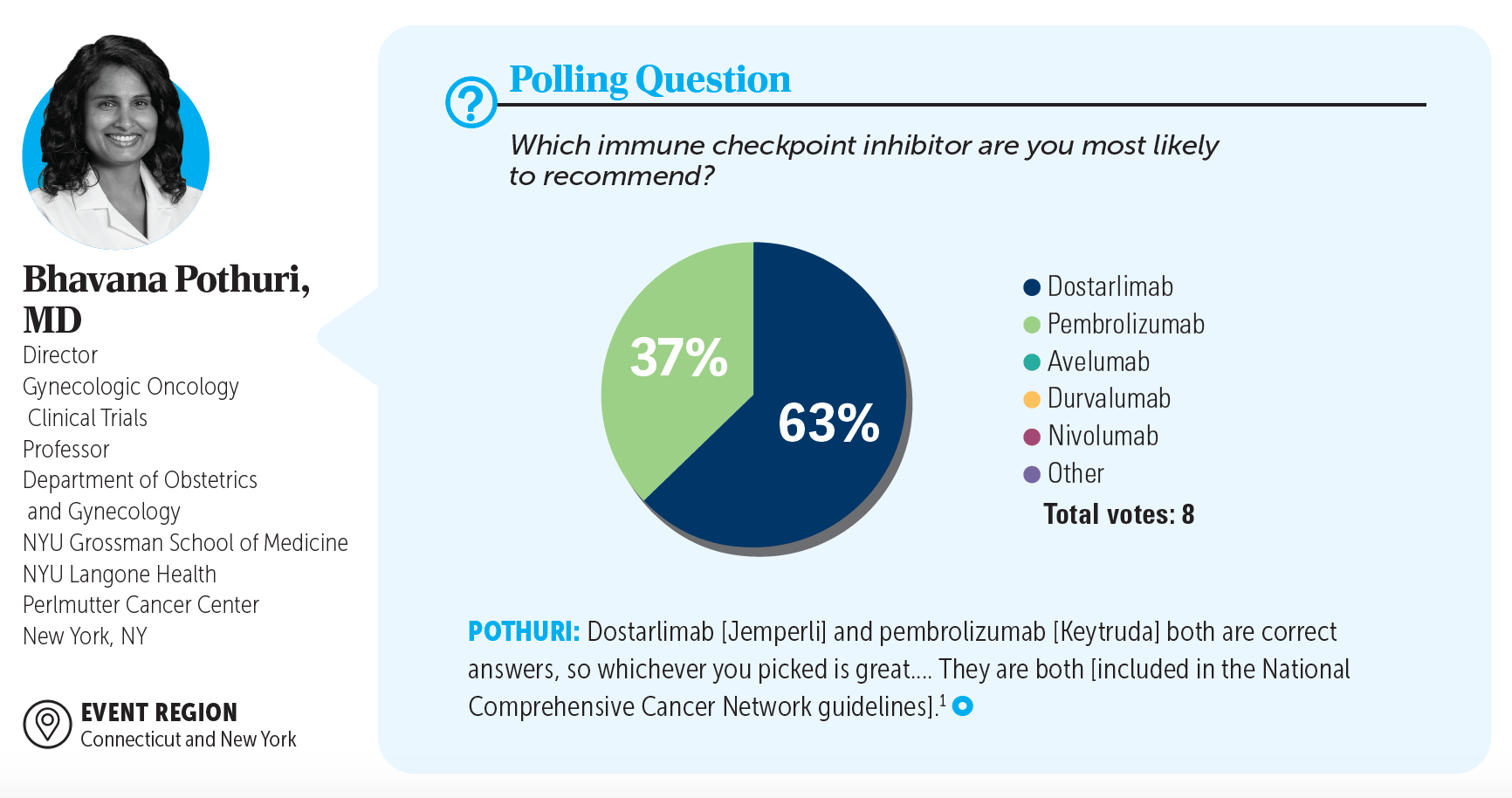
Fine-needle aspiration of the suprarenal mass confirmed metastatic endometrioid adenocarcinoma. The patient was counseled about systemic therapy options, during which she repeatedly expressed concerns about adverse events. Through shared decision-making, immune checkpoint inhibitor monotherapy was planned.

REFERENCE
1. NCCN. Clinical Practice Guidelines in Oncology. Uterine neoplasms, version 1.2024. Accessed February 2, 2024. http://tinyurl.com/28y356bt

Overcoming Barriers in Cancer Clinical Trials: A Path Forward for Better Patient Care
April 29th 2024Clinical trials play a pivotal role in developing effective therapies, yet their integration is challenged by issues such as insufficient reimbursement structures, misaligned incentives, physician burnout, and a complex regulatory environment.
Read More
Gholam Analyzes Treatment Outcomes for Advanced HCC in Child-Pugh B Population
April 28th 2024During a live Community Case Forum event in partnership with the Tennessee Oncology Practice Society, Pierre Gholam, MD, examined the current state of treatment for patients with hepatocellular carcinoma, looking in particular at what data is available for those with Child-Pugh B and C status who have poorer outcomes and have limited data from prospective clinical trials.
Read More