BRACAnalysis CDx Companion Diagnostic Progresses Personalized Ovarian Cancer Care
In mid-December of 2014, the FDA approved the BRACAnalysis CDx diagnostic test to accompany the use of Lynparza (olaparib) in advanced ovarian cancer.

Sharyn N. Lewin, MD
In mid-December of 2014, the FDA approved the BRACAnalysis CDx diagnostic test to accompany the use of Lynparza (olaparib) in advanced ovarian cancer.1,2The approval marked an important step forward in personalized treatment for patients withBRCA1- orBRCA2-positive mutations, who may benefit from treatment with olaparib. It also marked the first approval of a diagnostic test to accompany the use of a poly ADP-ribose polymerase (PARP) inhibitor in ovarian cancer.1,2
About Lynparza
Olaparib was approved as a monotherapy for patients with ovarian cancer who have received three or more lines of chemotherapy and have deleterious or suspected deleterious germline mutations in theBRCA 1orBRCA2genes.3,4This approval was granted under the FDA’s accelerated approval program based on an initially reported overall response rate of 34% and a median duration of response of 7.9 months, as observed in a single-arm phase II study of patients with germlineBRCA-mutated (gBRCAm) ovarian cancer.3,5Continued approval of this agent will therefore be contingent upon verification of its clinical benefit in phase III trials, which are ongoing.3,4
About BRACAnalysis CDx
Studies have found that approximately 15% of patients with epithelial ovarian cancer demonstrate positivity for mutations inBRCA 1orBRCA 2.6,7These findings have important clinical implications, because patients withgBRCAmhave better progression-free survival (PFS) and overall survival (OS) and improved response to certain treatments, including platinum-based therapies.6
BRACAnalysis CDX™ is an in vitro diagnostic test, which uses a blood sample to detect and classify variations (eg, single nucleotide variants, small insertions and deletions) in theBRCA 1andBRCA 2genes.8The testing is conducted at a single site, the Myriad Genetics Laboratory, and the results are used to identify patients with deleterious or suspected deleteriousgBRCAmwho are eligible for treatment with olaparib.8
BRCATesting Where Are We Now?
Late-breaking findings from Herzog et al reported at the 2015 Society of Gynecologic Oncology’s (SGO) Annual Meeting on Women’s Cancer have revealed a clearly suboptimal rate of testing forBRCAmutations among patients with ovarian cancer.9Since 2008, all patients with epithelial ovarian cancer have met the criteria for genetic testing, according to both National Comprehensive Cancer Network (NCCN) and SGO guidelines.9Despite this guidance, however, in a state-by-state analysis, only 27% of patients with ovarian cancer were tested forBRCA1/2mutations in 2013.9Although this represents a substantive increase from the 11.1% who were tested in 2008, the implication is that roughly three-quarters of patients with ovarian cancer are unaware of theirBRCAstatus. Importantly, even in those states with the highest testing rates (eg, Rhode Island, 44.4%), over 50% of patients remained untested within 1 year of diagnosis.9
On the potential for BRACAnalysis CDx to improve the rate of testing in patients with ovarian cancer, Johnathan M. Lancaster, MD, vice president of medical affairs at Myriad, commented to Targeted Oncology, “I’m highly optimistic that physicians in the community who are managing patients with ovarian cancer will recognize the potential benefit of being far more proactive about testing patients with a diagnosis of epithelial ovarian cancer for theirBRCAstatus… The irony is that testing all patients with epithelial ovarian cancer has been supported by NCCN guidelines and has been considered standard of care for a good number of years.”
Additional data on hereditary cancer testing for patients with ovarian cancer were presented at the SGO Annual Meeting by Lucy Langer, MD.10Langer reiterated the importance of identifying hereditary ovarian cancer caused by genes outside of BRCA1 and BRCA2. Overall, panel testing provided a 53.8% improvement in mutation detection over BRCA testing alone and led to the identification of mutations in genes that would not otherwise be suspected by clinical or family history alone. On the importance of panel testing Lancaster stated, "We understand the importance of a panel result for those patients who have a negative BRCA result and have created a path for patients to receive both an FDA-approved BRCA result as well as a panel for the same price as panel testing alone."
Implications for Ovarian Cancer Treatment
Sharyn N. Lewin, MD, director of gynecologic oncology at the Holy Name Medical Center, Regional Cancer Center, in Teaneck, New Jersey, and assistant clinical professor of obstetrics and gynecology at the Icahn School of Medicine at Mount Sinai Hospital in New York, is a national educator in hereditary genetics. Targeted Oncology spoke with Lewin about the BRACAnalysis approval and its implications for clinical practice. “When olaparib was given accelerated approval by the FDA in December, with BRACAnalysis CDx as a companion diagnostic, this was really a pretty historic, exciting, and important advance for those of us who are caring for women with ovarian cancer” Lewin said.
Figure 1. Percentage of newly-diagnosed patients with ovarian cancer tested in each state in 2013.(Mouseover to enlarge)
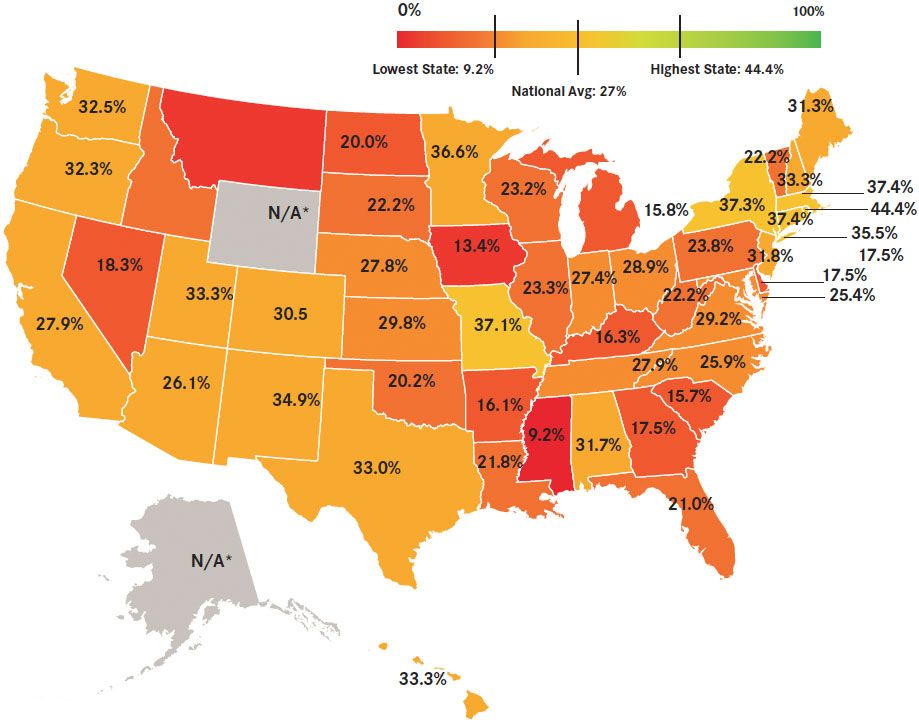
*<50 estimated new ovarian cancer case; percentage could not be calculated
Adapted from American Cancer Society. Cancer Facts & Figures:http://cancer.org/research/cancerfactsstatistics/allcancerfactsfigures/index. Accessed April 16, 2015.
In view of the late-breaking findings from this year’s SGO, Lewin encouraged more testing in this patient population. “That’s one of the exciting things about this FDA-approved testwe know that about 17% to 20% of unselected patients with ovarian cancer have aBRCAmutation, so that’s really a high percentage. And if we look at women with high-grade serous histology, almost 25% to 30% of those patients could have aBRCAmutation, so it’s a very high prevalence, yet less than 25% of women are identified and tested. So there’s really a huge gap and a lot of work to do in the medical profession to identify and test these women, especially with all of our societal guidelines, like ASCO, NCCN, and SGO. All of these guidelines say that women with ovarian cancer should be offered aBRCAtest.”
Lewin therefore sees the potential for this new diagnostic to encourage genetic testing in the ovarian cancer population. “The FDA has put forth a mandate that, in order to use this PARP inhibitor [olaparib], which has been very exciting in a lot of different arenas in which its been studied, mutations need to be detected by this FDA-approved test. This is just another reason why clinicians should be identifying and testing these patients, not only from a therapeutic standpoint, but also because these women are certainly at risk for secondary cancers, and their children and grandchildren are at risk for cancer, so anything that we can do to encourage clinicians to test patients is really important.”
Lancaster also sees the benefit of identifying these mutations early in the treatment course and recognizing the implications, not only in terms of hereditary risk, but also in terms of the potential to develop secondary cancers. “We now realize that patients with ovarian cancer who carry aBRCA 1or2alteration are known to have different responses to chemotherapy, and importantly, they’re known to have a longer survivalwhat that means is they have a longer time to develop a second malignancy such as a breast cancer.”
Both Lewin and Lancaster emphasized the importance of having an FDA-approved diagnostic method, and the extent of scientific rigor required for such an approval. “Not all laboratories are the same, and not all tests are the same…” Lewin said. “The majority of laboratories are certified by CLIA [Clinical Laboratory Improvement Amendments], which is completely different from an FDA-approved lab. Myriad’s BRACAnalysis CDx test was the first FDA-approved companion diagnostic, and the rigor that was required for FDA approval was unparalleled and unprecedented… second to none. I think that clinician’s don’t really understand what goes into thatthere’s so much validation that’s necessary that the FDA requires in terms of the test’s accuracy and reproducibility. All components of the assay have to be validated and no other lab and no other test for this gene has done that and can do that. And when it comes to a sensitive genetic test, validity, accuracy, and reproducibility are really crucial.”
Lancaster added, “The FDA, and ASCO, in fact, are clear in their position… the future of the art of molecular testing should be based upon an assurance to patients that the test they are receiving is of the highest quality, and of the highest caliber, and we believe that FDA approval represents that.”
Beyond its use in identifying patients for olaparib treatment, BRACAnalysis CDx also offers identifies and studies genetic variants, or uncharacterized polymorphisms, and informs patients about their potential clinical relevance (or lack thereof), which Lewin also sees as important. “No other lab has such a robust variance program… whenever you are doing genetic testing, you can have abnormalities in the protein that are called variants that may or may not have clinical significance, and Myriad is really the only laboratory that has a variants classification program, and these [variants] are constantly being reclassified as they are studied and tested, and patients will be notified that findings that may [initially] be identified as a variant could [in fact] be a benign finding in the future. And as a clinician, I think that’s what is really important to know.”
Lewin also believes the BRACAnalysis test will be more widely used as other PARP inhibitors are approved and begin to enter clinical use. “PARP [inhibitors] are probably the most exciting arena of personalized medicine when it comes to ovarian cancer, particularly for patients withBRCAmutations, but what’s kind of interesting about ovarian cancer is that there are a lot of ovarian cancer tumors that behave as though they were BRCA mutation [positive], even though the patient may not have aBRCAmutationand this is something that’s currently being studied in Europe—so there may be a lot more women with ovarian cancer that may benefit from PARP inhibitors than just those withBRCAmutations.”
Individual patients will gain from this type of mutational testing, according to Lewin. “They’ll know their genetic status, theirBRCAstatus, which is very important but it’s not only about therapeutic options with PARP inhibitors—we know that women withBRCAmutations and ovarian cancer live longer, and they do better on platinum-based drugs, particularly intraperitoneal platinum [therapy], so there are definitely therapeutic options that are really important for these womenbut also they can pass this on to their kids and grandkids, so just like with Angelina Jolie, which is so timely now, there’s a lot of opportunity for prevention with prophylactic surgery and increased surveillance. It’s important to know [BRCAstatus] for a lot of reasons, so that these cancers aren’t continued on into future generations.”
References
- Myriad Receives FDA Approval of BRACAnalysis CDx™ as Companion Diagnostic for Lynparza™(olaparib) in Ovarian Cancer Patients. Available at: http://www.myriad.com/investors/press-release-detail/?newsItemId=888523 Accessed: March 31, 2015.
- FDA approves Lynparza to treat advanced ovarian cancer. Available at: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm427554.htm Accessed: March 31, 2015.
- LYNPARZA™ approved by the US food and drug administration for the treatment of advanced ovarian cancer in patients with germline BRCA-mutations. Available at: http://www.astrazeneca.com/Media/Press-releases/Article/20141219--lynparza-approved Accessed: March 31, 2015
- Lynparza ™ Prescribing Information. December 2014. AstraZeneca Pharmaceuticals LP. Wilmington DE.
- Kaufman B, Shapira-Frommer R, Schmutzler RK, et al. Olaparib monotherapy in patients with advanced cancer and a germline BRCA1/2 mutation.J Clin Oncol. 2015;33(3):244-250.
- Alsop K, Fereday S, Meldrum C, et al. BRCA mutation frequency and patterns of treatment response in BRCA mutationpositive women with ovarian cancer: A report from the Australian Ovarian Cancer Study Group.J Clin Oncol. 2012;30(21):2654-2663.
- Pal T, Permuth-Wey J, Betts JA, et al. BRCA1 and BRCA2 mutations account for a large proportion of ovarian carcinoma cases.Cancer. 2005;104(12):2807-2816.
- BRACAnalysis CDx™ Technical Information Summary. www.myriad.com. Accessed March 30, 2015
- Herzog T, Saam J, Arnell C, et al. A State by state analysis of BRCA1 and BRCA2 testing in patients with ovarian cancer. Poster presented at: Society for Gynecologic Oncology Meeting, March 28-31, 2015.