Momelotinib Addresses Anemia in Myelofibrosis
Findings from the MOMENTUM study evaluating momelotinib demonstrated symptom, spleen, and anemia benefits in myelofibrosis compared with danazol in previously treated patients with anemia and intermediate- or high-risk myelofibrosis.
John O. Mascarenhas, MD
Professor of Medicine
Icahn School of Medicine at Mount Sinai
New York, NY

With the approval of momelotinib (Ojjaara) for patients with myelofibrosis, oncologists and hematologists now have 4 JAK inhibitors for consideration to treat the disease. Determining the appropriate agent and the appropriate timing during the course of the disease to initiate treatment requires expertise and nuance and was the focus of a presentation by John O. Mascarenhas, MD, during the 41st Annual Chemotherapy Foundation Symposium Innovative Cancer Therapy for Tomorrow® conference.1
National Comprehensive Cancer Network guidelines suggest stratifying patients by symptom burden and platelet counts.2 In patients whose platelet counts are less than 50 × 109/L, and who are transplant eligible, allogeneic hematopoietic stem cell transplant (HSCT) remains the only curative approach.
For patients who are transplant ineligible, enrollment in a clinical trial or treatment with pacritinib (Vonjo) are options, according to the guidelines.
In patients whose platelet counts are greater than or equal to 50 × 109/L, if the patient is transplant ineligible, ruxolitinib (Jakafi), fedratinib (Inrebic), pacritinib, and momelotinib (Ojjaara) are JAK inhibitors for consideration, as is enrollment in a clinical trial. If the patient is transplant ineligible and has symptoms of anemia, treatment focuses on managing the myelofibrosis- associated anemia.2
Findings from the MOMENTUM study (NCT04173494)3 evaluating momelotinib demonstrated symptom, spleen, and anemia benefits in myelofibrosis compared with danazol in previously treated patients with anemia and intermediate- or high-risk myelofibrosis.
A total of 195 patients were randomly assigned 2:1 to receive either momelotinib 200 mg and placebo vs danazol 600 mg and placebo. Patients were allowed to enroll in open-label crossover if they had confirmed progression after week 24.
The primary end point was the Myelofibrosis Symptom Assessment Form (MFSAF) total symptom score (TSS) response rate at week 24 (defined as ≥ 50% reduction in mean MFSAF TSS over the 28 days immediately before the end of week 24 compared with baseline). Topline findings are presented in the TABLE.3
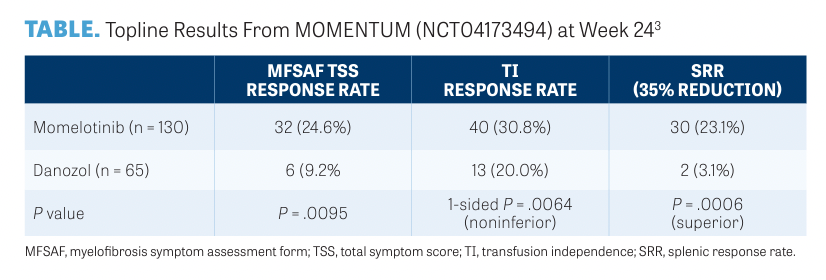
The most frequent grade 3 or higher treatment-emergent adverse events (TRAEs) with momelotinib and danazol were hematological abnormalities: anemia (61% vs 75%) and thrombocytopenia (28% vs 26%). Most frequent non-hematological grade 3 or higher TRAEs were acute kidney injury (3% vs 9%) and pneumonia (2% vs 9%).
“An interesting finding here is that patients who had achieved transfusion independence had durable responses,” Mascarenhas said.
Mascarenhas provided an overview of his myelofibrosis treatment approach, which excluded transplant. In the first line, patients who have a platelet count less than 75 to 100 × 109/L are candidates for pacritinib. Patients with a count of greater than 100 × 109/L should be treated with ruxolitinib or fedratinib, followed by a clinical trial. Patients who are red blood cell transfusion dependent are candidates for momelotinib, followed by a JAK inhibitor and erythropoiesis-stimulating agents or luspatercept-aamt (Reblozyl).
In the second line, patients with a platelet count less than 100 × 109/L are candidates for pacritinib. In patients experiencing splenomegaly, fedratinib should be considered; in patients with anemia, momelotinib is the appropriate treatment choice.
In the case of ruxolitinib failure, a JAK inhibitor-based combination therapy should be considered as well. If anemia arises after combination therapy, the clinician can consider an erythropoiesis-stimulating agent, immunomodulating drugs, or androgen therapy.
In advanced stages, clinicians can choose from DNA methyltransferase inhibitors, isocitrate dehydrogenase inhibitors, or BCL2 inhibitors.
“In summary, JAK inhibitors afford patients with spleen, symptom, and cytopenia benefit but do not achieve remission,” Mascarenhas said. “Second-line options after ruxolitinib failure include fedratinib, pacritinib, and now momelotinib. In the future, we will likely see a shift to upfront combination therapy, non JAK2 inhibitor agent sequencing, and increase in survival,” Mascarenhas concluded.
REFERENCES
1. Mascarenhas JO. Effectively integrating JAK inhibitor options for the management of myelofibrosis. Presented at: 41st Annual Chemotherapy Foundation Symposium; November 8-10, 2023; New York, NY.
2. NCCN. Clinical Practice Guidelines in Oncology. Myelodysplastic syndromes, version 3.2023. Accessed November 28, 2023. https://tinyurl.com/43tmz8ew
3. Verstovsek S, Gerds AT, Vannucchi AM, et al. Momelotinib versus danazol in symptomatic patients with anaemia and myelofibrosis (MOMENTUM): results from an international, double-blind, randomised, controlled, phase 3 study. Lancet. 2023;401(10373):269-280. doi:10.1016/S0140-6736(22)02036-0
Overcoming Barriers in Cancer Clinical Trials: A Path Forward for Better Patient Care
April 29th 2024Clinical trials play a pivotal role in developing effective therapies, yet their integration is challenged by issues such as insufficient reimbursement structures, misaligned incentives, physician burnout, and a complex regulatory environment.
Read More
Gholam Analyzes Treatment Outcomes for Advanced HCC in Child-Pugh B Population
April 28th 2024During a live Community Case Forum event in partnership with the Tennessee Oncology Practice Society, Pierre Gholam, MD, examined the current state of treatment for patients with hepatocellular carcinoma, looking in particular at what data is available for those with Child-Pugh B and C status who have poorer outcomes and have limited data from prospective clinical trials.
Read More